RABYD-VAX combines the expertise of five European research institutes to develop and validate a vaccine that protects against both rabies and yellow fever/Japanese encephalitis virus and which could ultimately be given as a prophylactic childhood vaccine incorporated in the standard childhood vaccination schedule of endemic regions. This side-by-side with domestic and wildlife animal vaccination programmes to eradicate rabies. This vaccine is based on a novel proprietary vaccine technology developed by the team of the coordinator (KU Leuven) and has several significant advantages over the currently used commercial vaccines. Within this consortium we aim to ready this vaccine candidate for the late pre-clinical development stage.
Why rabies?
Rabies is a devastating and massively neglected disease for which the mortality rate and burden per capita falls disproportionately upon the poorest regions of the world. Rabies is a viral disease that is largely transmitted to humans via a bite by infected animals. Once the first clinical symptoms (such as hydrophobia) have developed, the disease is uniformly lethal, and patients, if not put into an artificial coma, die in great agony. Worldwide, rabies causes approximately 58.000 deaths from which the majority are young children. A vaccine exists but is associated with several drawbacks such as the need for a cold chain (kept at cool temperature), high costs, multiple dosing regimen with concomitant visits to health care facilities, etc. due to which the coverage remains unacceptably low.
Why yellow fever and Japanese encephalitis?
Depending on the region of application we will develop a bivalent rabies/yellow fever or rabies/Japanese encephalitis vaccine (see below ‘technology’).
The yellow fever virus (YFV), a mosquito-borne flavivirus, causes severe and life-threatening infections with jaundice, systemic bleeding, shock and multi-organ failure. An estimated 900 million people living in 45 endemic countries of Africa and Latin America are at high risk of infection. Although safe and highly efficient live-attenuated prophylactic vaccines [YFV-17D, Stamaril® and YF-Vax®] are available, an estimated 200,000 cases of yellow fever still occur annually, resulting in ~30,000 deaths (www.who.int) because of inadequate supplies, the need for trained staff and a cold chain.
The Japanese encephalitis virus (JEV), also a mosquito-borne flavivirus, causes viral encephalitis in many countries of Asia, with an estimated 68,000 clinical cases every year. The case-fatality rate among the patients that develop encephalitis is up to 30%, permanent neurologic or psychiatric sequelae is reported in 30-50%. As for rabies, it is primarily a children’s disease. More than 3 billion people are at risk of infection.
Which technology?
The team of Prof. Neyts (coordinator) developed a novel vaccine platform technology, named PLLAV, i.e. plasmid-launched live-attenuated vaccine [patent WO/2014/174078, inventors K. Dallmeier & J. Neyts]. In essence, the technology consists of a proprietary Bacterial Artificial Chromosome (BAC) shuttle vector in which the genome of the yellow fever virus vaccine YFV-17D, which is a live-attenuated vaccine (LAV), has been cloned. Once this recombinant DNA plasmid is introduced in an eukaryotic cell, the LAV is expressed and immunity triggered. Hence, this approach combines, (1) the high efficacy of LAVs for inducing protective immunity and (2) the physical and genetic stability of classical DNA vaccines, bypassing the manufacturing problems and need for a cold chain. Moreover, and as outlined below, this approach allows to conveniently design and rapidly produce vaccines against other pathogens (i.e. rabies) as well. The PLLAV technology presents numerous key advantages over the currently available vaccines, i.e. (i) it can be produced at high quantities in fermenters without the need for cell cultures or embryonated chicken eggs; (ii) it’s genetically stable; (iii) no cold-chain is needed and (iv) it can be administered needle-free. (see PLLAV concept for more information)
In fact the PLLAV technology addresses the need for good vaccines for the developing world as articulated by Médecins Sans Frontières “developing vaccines that are better adapted to reach children in remote or unstable locations – vaccines that do not require refrigeration, do not require needles, and that can be given in fewer doses […], paying enough attention to getting vaccine prices down”.
5 objectives
Within this four-year during project we will focus on the following five key objectives:
- Objective 1 – development of PLLAV-rabies vaccine candidate
- Objective 2 – study of the immunity induced by PLLAV-rabies vaccines
- Objective 3 – dual protection from lethal vaccine challenge
- Objective 4 – proof-of-concept study for immunogenicity and safety
- Objective 5 – development trajectory, transition to advanced pre-clinical development
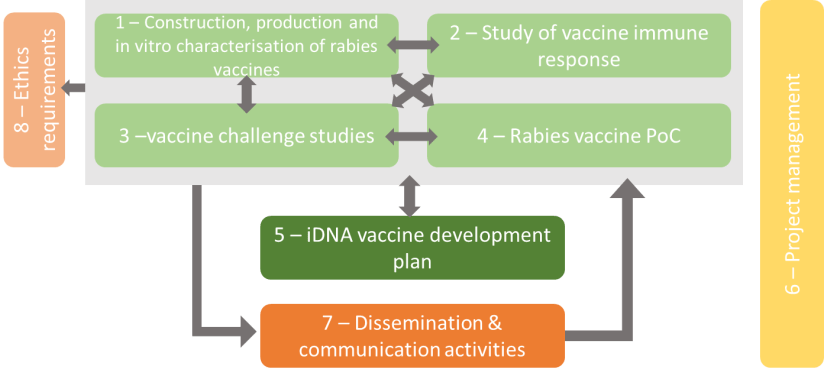
8 WPs
To meet its five key objectives, the scientific project has been organized in 8 Work Packages, with:
- WP1-4 dedicated to basic research
- WP5 to the development of a well-based vaccine development plan
- WP6 to management
- WP7 to dissemination and communication activities
- WP8 to supervision of ethic requirements